The disorder and non-uniform of Pt clusters are interesting to explore. In this study, we focus on clarifying the nature of the strong interaction between platinum nanoparticles and GNS. It means the particle size and morphology may affect CO species bonding on clusters sites 34.Ĭommonly, the particle size, shape, chemical composition, metal–support interaction, metal–reactant/solvent interaction and different size (single atoms, nanoclusters, and nanoparticles) of metal exhibits different performance on the catalytic properties of metal catalysts 35. In addition, the small clusters may cause break the scaling relationship in catalysis and may exceed the intrinsic limit on catalytic activity 33. Noteby, smaller nanoparticles generally have many weaknesses those are reducing lifetime in electrochemical devices weak interaction between platinum and carbon black, resulting nanoparticle dissolution aggregation and detachment, leading to PEFC performance degradation and carbon corrosion and associated loss of ECSA over time at high cathode potentials 32. The reducing Pt nanoparticles size tends to increasing electrochemical surface area (ECSA) and decreases loading. Pt nanoparticles have been applied as catalysts on fuel cells 31. Graphene as well as a supporting material may assist to homogenize and disperse noble metals 26, 27, to generate metal clusters 28, reducing CO oxidation reaction by assisting strong interactions between support–cluster 29, and protecting Pt cores on fuel cells operating system 30. Their electronic properties and catalytic activity are tuned by the interaction between the central metal and the neighboring surface atoms, and their atomically dispersed nature allows for metal utilization of up to 100% 25. The nanoscales materials may be designed referring to controllable transportation, separation, classification of nano-objects 23 and type of supporting materials 24. However, the metal loading density must be kept low (usually below 0.5%) to avoid the formation of metal nanoparticles through sintering 22. The interaction between metal–support may induce charge transfer between metal and support, tame electronic structure of supported metals, impact adsorption energy of reaction intermediates, and eventually change the catalytic performance 20, 21.

The shape, direction, size and alloy of Pt may affect the ORR catalytic activity and durability 16, 17, 18, 19. The other groups reported Mg clusters in 3D hierarchical structure (3DHS) have a superior cycling stability of up to 1350 h 15.

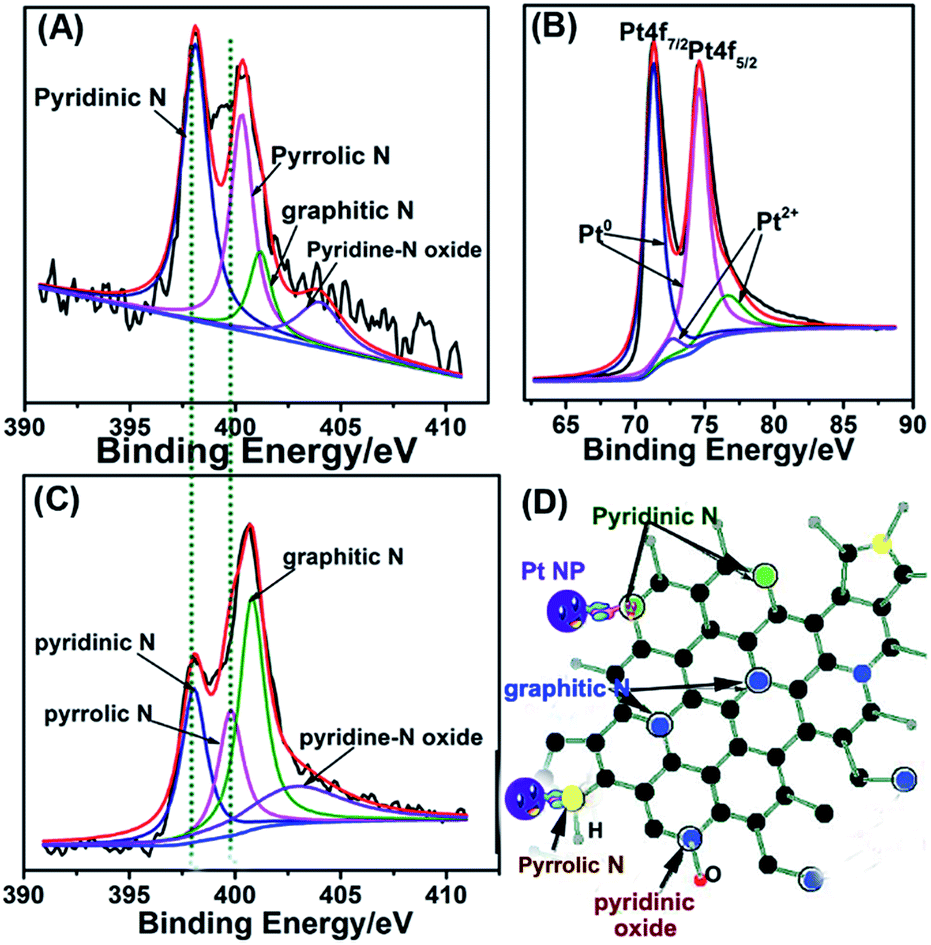
That is caused size and shape of Pt clusters may affect the properties of interaction between Pt clusters and graphene sheets 14. In the case of Pt 10–70 wt.%/GNS, they perform the outstanding properties and Pt Subnano clusters were exist on GNS where those are never be found on the other carbon supporting materials 2, 3. Interestingly, in order to improve properties of GNS, it may be modified by using N-doping to generate N-GNS 11, 12, 13. Fortunately, GNS also may be synthesized with large scale and facile method 9 and it shows the remarkable properties as a support material 10. They also have the outstanding properties in term of catalytic activity and chemically interaction properties 2, 3, 4, 5, 6, 7, 8. Previously, we found that Pt Subnano clusters were well deposited on Graphene Nano Sheets (GNS). Carbon has been recognized as efficient metal-free electrocatalysts for ORR and OER in fuel cells and batteries, such as 2D graphene and 3D graphite 1.
